Smarter Approaches to Software Validation in Life Sciences
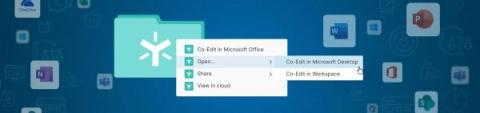
Global regulations regarding software validation (e.g., 21 CFR Part 11, Annex 11, etc.) have been in place for many years. With an increasing number of software-as-a-service vendors offering GxP-compliant solutions, it is more important than ever that CSV and QA teams of all sizes make use of sensible approaches to software validation to better manage their overhead while maintaining compliance with regulatory requirements.